Immune cells can be used to develop cell therapies for cancer treatment
Recent advances in cancer treatment have unleashed the power of immune cells, enabling them to recognize and kill tumors. Cell therapies are built from these powerful immune cells using genetic engineering to improve their therapeutic activity. One of the most widely adopted forms of cell therapy uses a particular type of immune cell called a T cell, and the FDA-approved versions of these therapies are called CAR-T cell therapies.
Autologous CAR-T cell therapies use a genetically engineered version of a patient’s own T cells, which after genetic engineering, are infused back into the patient to treat hematologic malignancies, or cancers of the blood. While these therapies have been approved by the FDA for the treatment of hematologic malignancies, these autologous CAR-T cell therapies are cumbersome and slow to make, often requiring three to four weeks to manufacture. Autologous CAR-T cell therapies have also been associated with severe adverse events such as cytokine release syndrome and neurotoxicity. They represent the earliest generation of an emerging therapeutic class of engineered immune cells.
Natural killer (NK) cells are a vital component of the immune response to cancer
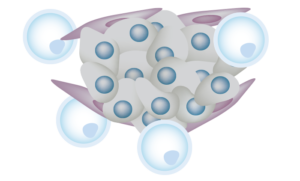
Natural killer (NK) cells are a type of human immune cell that plays a critical role as the first line of defense against tumor cells and virally infected cells. NK cells, as part of the body’s innate immune system, possess the unique ability to attack cells that do not display normal surface markers. NK cells play an essential role in immune surveillance of tumors by distinguishing between malignant and healthy cells and recruiting other immune cells to bolster anti-tumor activity. When activated, NK cells secrete molecules that can either directly break down tumor cells or activate the apoptosis cell death program in tumor cells.
Engineered NK cell therapies overcome the limitations of T cell therapy for cancer
 Cell therapies hold enormous potential as transformative treatments for cancer, as shown by the clinical success of autologous CAR-T therapies in hematologic cancers. However, these therapies have been limited to patients with hematologic malignancies. Furthermore, current autologous CAR-T cell therapies are inefficient to manufacture and have significant safety concerns; both of which create a high cost to the healthcare system and a significant burden to patients and their families. These characteristics have limited the application of these therapies primarily to academic medical centers.
Cell therapies hold enormous potential as transformative treatments for cancer, as shown by the clinical success of autologous CAR-T therapies in hematologic cancers. However, these therapies have been limited to patients with hematologic malignancies. Furthermore, current autologous CAR-T cell therapies are inefficient to manufacture and have significant safety concerns; both of which create a high cost to the healthcare system and a significant burden to patients and their families. These characteristics have limited the application of these therapies primarily to academic medical centers.
CAR-NK cell therapies have the potential to overcome the challenges of earlier autologous CAR-T cell therapies and deliver transformative treatments to patients. Allogeneic CAR-NK cell therapies can be derived from healthy donors, engineered to improve activity, and stored. These features enable off-the-shelf products manufactured from one donor that are ready to be infused into multiple patients at the time of diagnosis. Specifically, allogeneic CAR-NK cell therapies evaluated in early clinical trials do not cause GvHD, cytokine release syndrome or neurotoxicity while maintaining a high degree of efficacy in difficult-to-treat hematologic malignancies.
Catamaran’s CAR-NK cell therapies expand the potential of cell therapy to reach patients with solid tumors
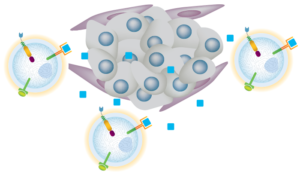 Historically, CAR-T cell therapies have only demonstrated meaningful efficacy for hematologic malignancies. Catamaran’s CAR-NK cell therapies offer the possibility of highly potent, off-the-shelf cell therapies for the treatment of solid tumors and other cancers, without the safety issues associated with T cell-based therapies. The first generation of cell therapies have been successful in hematological malignancies but have encountered challenges in solid tumors. Like most immune cells, our body’s NK cells are suppressed by multiple factors and barriers generated by solid tumors and the surrounding tumor microenvironment (TME). Using the TAILWIND® platform, Catamaran’s scientists are engineering CAR-NK cell therapies with optimized CAR architecture for improved cell activation and potency-boosting switches to address the hostile TME. This approach opens the possibility that Catamaran’s cell therapies will transform the treatment of cancer by reaching more patients and more cancer types.
Historically, CAR-T cell therapies have only demonstrated meaningful efficacy for hematologic malignancies. Catamaran’s CAR-NK cell therapies offer the possibility of highly potent, off-the-shelf cell therapies for the treatment of solid tumors and other cancers, without the safety issues associated with T cell-based therapies. The first generation of cell therapies have been successful in hematological malignancies but have encountered challenges in solid tumors. Like most immune cells, our body’s NK cells are suppressed by multiple factors and barriers generated by solid tumors and the surrounding tumor microenvironment (TME). Using the TAILWIND® platform, Catamaran’s scientists are engineering CAR-NK cell therapies with optimized CAR architecture for improved cell activation and potency-boosting switches to address the hostile TME. This approach opens the possibility that Catamaran’s cell therapies will transform the treatment of cancer by reaching more patients and more cancer types.

